
As of August 7, 2020, the US Centers for Disease Control and Prevention (CDC) has received reports of 640 confirmed cases of Salmonella Newport infections across 43 states.
Eight-five outbreak victims have required hospitalization.
In addition to the US outbreak, 239 confirmed cases have been reported to the Public Health Agency of Canada (PHAC) across seven provinces as of August 7th. Twenty-nine Canadians have been hospitalized.
According to the US Food and Drug Administration (FDA) illnesses in both countries have been linked to red onions grown and supplied by Thomson International, Inc. (Thomson) of Bakersfield, CA.
On August 1st, Thomson recalled all varieties of onions that could have come in contact with potentially contaminated red onions, including red, yellow, white, and sweet yellow onions shipped from May 1, 2020 to present.
The recalled onions were supplied to customers in the USA and Canada under several brand names, including:
- Thomson Premium
- TLC Thomson International
- Tender Loving Care
- El Competitor
- Hartley’s Best
- Onions 52
- Majestic
- Imperial Fresh
- Kroger
- Utah Onions
- Food Lion
In addition to the Thomson recall, companies in the United States and Canada have recalled Thomson-supplied onions or products that contain onions on the Thomson recall list. Please follow the links to the individual recall notices for a complete list of products.
US recalls
ALDI
Brookshire’s Food and Pharmacy (Southwest Turkey Cobb Salads)
Costco Wholesale (multiple recall notices)
Department of Defense Commissaries
Giant Eagle
Food Lion
Fred Meyer
Fry’s Food Stores
H-E-B
Kroger
Martin’s Groceries, Supermarket & Pharmacy
Publix Super Markets
Schnucks
Smith’s
Stop&Shop
Super 1 Foods
Taylor Farms (products without meat)
Taylor Farms (products containing meat)
Walmart (multiple recall notices)
Canadian recalls
List of recalled products in Canada
Costco Canada
Freshpoint Foodservice
Giant Tiger
Multiple companies
Sysco
This outbreak is not over. Both CDC and PHAC are anticipating additional cases. To keep yourself and your family and your customers safe, please take note of the following guidance:
Advice to consumers
Check your home for red, white, yellow, and sweet varieties, including whole, sliced, or chopped onions, and any prepared foods that contain onions as an ingredient, such as premade salads, sandwiches, wraps, salsas or dips.
- If you have onions at home:
- Look for a label showing where the onion was grown. It may be printed on the package or on a sticker.
- If the packaging or sticker shows that it is from Thomson International Inc., don’t eat it. Throw it away and wash your hands.
- If it isn’t labeled, don’t eat it. Throw it away and wash your hands.
- If you don’t know whether the onion found in a premade salad, sandwich, wrap, salsa or dip contains onions from Thomson International Inc., don’t eat it. Throw it away and wash your hands.
- Wash and sanitize any surfaces that may have come in contact with onions or their packaging, such as countertops, fridge drawers, pantry shelves, knives, and cutting boards.
- If you buy onions at grocery or convenience stores:
- Make sure they are not selling onions from Thomson International Inc., or serving fresh foods prepared with them.
- If you can’t confirm that the onion in stores is not from Thomson International Inc., don’t buy it.
- If you order salad or any other food items containing onions at a restaurant or food establishment:
- Ask the staff whether their onions come from Thomson International Inc. If they did, or they don’t know, don’t eat it.
- Do not eat any recalled food products. Check to see if you have recalled food products at home. If you do, throw them out and wash your hands.
- If you believe you are experiencing symptoms of a Salmonella infection, consult a healthcare practitioner immediate.
- If you have been diagnosed with a Salmonella infection or any other gastrointestinal illness, do not cook food for other people.
- Contact your local public health authority to report any food safety concerns at restaurants or grocery stores, or if you suspect food poisoning from a restaurant or other food establishments.
Advice to restaurants, retailers, suppliers and distributors
- Check the label on bags or boxes of onions, or ask their suppliers about the source of their onions.
- Do not ship or sell onions from Thomson International Inc. of Bakersfield, California, USA, or any products made with these onions.
- Clean and sanitize all surfaces and storage bins that onions may have come in contact with, including cutting boards, countertops, slicers, utensils, and containers used to store or transport them.



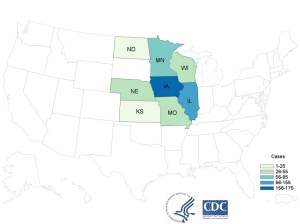 The Cyclospora outbreak in the US has spread to 8 midwestern states, and has sickened at least 509 individuals as of
The Cyclospora outbreak in the US has spread to 8 midwestern states, and has sickened at least 509 individuals as of