This story by Bill Marler first appeared on the Marler Blog and is reposted here with the author’s permission.
On August 19, 2024, four-year-old Noah Ruiz experienced onset of diarrhea. A stool specimen collected at St. George Regional Medical Center on 8/22/2024 was PCR positive for E. coli O157:H7. The Utah Department of Health (UDH) State Public Health Laboratory (SPHL) isolated E. coli O157:H7 in Noah’s specimen (Accession Number 2017861). UDH SPHL conducted whole genome sequencing (WGS) on Noah’s isolate. Results were assigned identification numbers PNUSAE198092/SAMN43527540 and Allele code EC1.1-1.1.259.1566. Noah developed Hemolytic Uremic Syndrome and spent a month at Intermountain Health Primary Children’s Hospital.
Public health investigators interviewed Noah’s parents about potential exposures to E. coli O157:H7. Noah’s food history was unremarkable. He did not consume ground beef, leafy greens, sprouts, or fresh herbs. Investigators noted that Noah did have close contact with his dog who was fed Darwin’s Raw Pet Food. The dog, who exclusively consumed Darwin’s pet food, began vomiting one day before Noah became ill. The dog’s symptoms resolved without veterinary care. Noah’s parents reported that Noah had direct contact with the dog but had no known direct contact with the dog food.
Raw pet food, like other uncooked animal products, is not subjected to a validated kill step to eliminate pathogenic microorganisms prior to distribution. As a result, it may harbor dangerous pathogens such as Salmonella, Listeria monocytogenes, and pathogenic Escherichia coli. These organisms can persist in raw meat and contaminate surfaces, feeding utensils, and the surrounding environment during routine handling and feeding.
Pets that consume contaminated raw food can become infected and may shed pathogens in their saliva and feces, even if they appear clinically healthy. This creates multiple avenues for transmission within the household. Children are particularly at risk because they frequently engage in close contact with pets, including touching, petting, and exposure to contaminated surfaces. Transmission can occur through direct contact with the animal, contact with contaminated household environments, or improper handling of the raw product itself.
Vulnerable populations—including young children, the elderly, pregnant women, and immunocompromised individuals—face an elevated risk of severe illness when exposed to pathogenic E. coli and other foodborne pathogens. Infections may result in serious complications, including hemolytic uremic syndrome (HUS), a potentially life-threatening condition. Given these risks, the handling and use of raw pet food products presents a foreseeable and preventable public health hazard, particularly in households with high-risk individuals.
In May 2025, Noah’s parents provided unopened frozen packages of Darwin’s pet food for testing to IEH Laboratories & Consulting Group, a private laboratory accredited by the FDA Laboratory Accreditation for Analyses of Foods (LAAF) program. IEH Laboratories isolated E. coli O157:H7 in a sample of Darwin’s Beef Dog Food (Lot 10662). In addition, one sample of Darwin’s Chicken Dog Food tested positive for Salmonella Infantis (Lot 10683), and one sample of Darwin’s Duck Dog Food (Lot 10638) tested positive for Salmonella Hadar. IEH Laboratories & Consulting Group conducted WGS on an E. coli O157:H7 isolate obtained in a sample of Darwin’s Natural Pet Product Biologics Beef Recipe for Dogs. WGS results were assigned CFSAN142494 and posted on the National Center for Biotechnology Information. The E. coli O157:H7 isolated in the dog food was a genetic match to the E. coli O157:H7 isolated in Noah Ruiz.

On July 7, 2025, Noah’s father, Fred Ruiz, was interviewed by FDA Investigator, Antonetta M. Colacchio. Fred summarized in a sworn statement dated July 8, 2025, that the family dog, Rodeo, had been fed Darwin’s Natural Pet Products for ten years. He provided Investigator Colacchio with Darwin’s Natural Pet Products order confirmations dated June 14, 2024, and August 9, 2024. He described how the product was delivered frozen to his home and immediately placed in the freezer. Freezer space was dedicated to just holding the frozen pet food. A package of frozen dog food was placed in a dedicated open plastic container and set to thaw in their refrigerator. Rodeo was fed in the laundry room. Immediately after feeding, the food bowl was rinsed and placed in the dishwasher to sanitize. Fred said that his children, Noah and Roman, were aware and taught not to touch or handle any of the raw pet food. They were not allowed to help feed Rodeo. Rodeo did not eat any other food or treats. Fred also stated that Noah and Roman would cuddle and play with Rodeo all the time and that Rodeo would occasionally lick his sons’ faces when they interacted.
On July 29, 2025, the FDA issued an advisory cautioning pet owners that they should not feed certain lots of Darwin’s Natural Pet Products Pet Food due to contamination with E. coli O157:H7 and Salmonella. FDA recommended that Arrow Reliance Inc., the maker of Darwin’s Natural Pet Food Products, recall the product lots that tested positive for foodborne pathogens. At the time the advisory was issued, the firm had not recalled the affected products. The advisory noted that the products, which were manufactured in May or June 2024, were sold frozen, had no expiration date on the label, and could still be in consumers’ freezers. The FDA advised consumers to check their freezers for the affected lots of Darwin’s Natural Pet Products prior to feeding their pets.
The FDA noted:
- The FDA is cautioning pet owners that a sample of Darwin’s Natural Pet Products beef dog food made by Arrow Reliance, Inc. tested positive for Escherichia coli (E. coli) O157:H7 that is associated with a human illness. In addition, one sample of Darwin’s chicken dog food and one sample of Darwin’s duck dog food tested positive for Salmonella Infantis and Hadar, respectively. Closed product from the three affected lots, which were obtained from the household freezer of the ill human, was tested by a private third-party laboratory in May and June 2025.
- In August 2024, a four-year-old child became infected with E. coli O157:H7 and subsequently developed hemolytic uremic syndrome (HUS), requiring hospitalization and extensive follow-up care. Whole genome sequencing (WGS) provided by the third-party laboratory shows that the E. coli O157:H7 present in the closed product sample of the Darwin’s beef dog food from the household freezer is a genetic match to the E. coli O157:H7 detected in a stool sample from the ill child.
- According to complaint information provided to the FDA, a dog in the household that exclusively ate Darwin’s pet food began vomiting one day before the child fell ill. The dog’s symptoms resolved without veterinary care.
- The child had direct contact with the family dog but no known direct contact with the contaminated dog food, suggesting that the E. coli O157:H7 exposure may have been via contact with the family dog or through objects or surfaces in contact with the waste of the dog that ate the food.
- Because this incident occurred in 2024, there were no retail samples available for collection and testing when the complaint information and private laboratory results were submitted to the FDA in June 2025. Additionally, Darwin’s Natural Pet Products does not have samples from the same product lot available for additional testing. Therefore, the FDA reviewed the results of the third-party testing conducted by the lab, which is accredited by the FDA Laboratory Accreditation for Analyses of Foods (LAAF) program.
- Darwin’s Natural Pet Products are sold online through a subscription service.
- The FDA recommended that Arrow Reliance, Inc. recall the product lots that tested positive for E. coli O157:H7 and Salmonella. To date, the firm has not recalled the affected products. These products, which were manufactured in May or June 2024, were sold frozen, have no expiration date on the label, and could still be in consumers’ freezers. Therefore, the FDA advises consumers to check their freezers for the affected lots of Darwin’s Natural Pet Products prior to feeding to pets.
- If consumers have any pet food on the list below, they are advised to throw it away in a secure container. Do not feed it to pets. Do not donate the food.
In summary, IEH Laboratories & Consulting Group, an FDA accredited laboratory, isolated E. coli O157:H7 in an unopened package of Darwin’s Natural Pet Products pet food obtained from the Ruiz home. The isolate was a genetic match by WGS to an E. coli O157:H7 isolate obtained from Noah Ruiz. These results prove beyond reasonable doubt that Noah Ruiz acquired his E. coli O157:H7 infection as a result of his exposure to Darwin’s Natural Pet Products.
The precise transmission pathway by which Noah Ruiz acquired the E. coli infection, however, cannot be definitively determined. While direct contact with Rodeo or Darwin’s raw pet food is one viable route, it is equally plausible that exposure occurred through any number of indirect and environmental pathways. These include, but are not limited to, contact with liquids or juices from the raw product; contaminated packaging; surfaces onto which such packaging was placed; and cross-contamination during handling or disposal, including contact with household trash. Additional potential vectors include kitchen and food-preparation surfaces—such as countertops, sinks, faucets, and handles–as well as contaminated utensils, including pet bowls, sponges, and cleaning cloths. Once introduced, pathogens like E. coli can persist and spread throughout the broader kitchen environment—including floors, tables, and “splash zones”—and beyond.

The risk of environmental contamination is not confined to the kitchen. Rodeo was fed in the family’s laundry room, his food was thawed in the household refrigerator, and his bowl was cleaned in the family dishwasher. These practices created multiple potential routes of transmission between pet-related items and human food items, utensils, and shared household surfaces.
The evidence further establishes that Rodeo had been fed Darwin’s raw pet food exclusively since a very young age, resulting in the continuous and repeated introduction of raw animal product into the home environment. This was an ongoing condition that increased the likelihood of environmental contamination over time.
Young children are particularly vulnerable to indirect exposure pathways. They frequently crawl, play on floors, and place their hands or objects in their mouths. Contaminants present on floors and other household surfaces can be readily spread through foot traffic and normal movement within the home, significantly expanding the scope of exposure beyond the initial point of contamination.
Presence and Regulation of Pathogens in Animal Foods
The U.S. Food & Drug Administration (FDA) monitors biological contaminants in animal food, including E. coli, Salmonella, and Listeria monocytogenes. As the FDA notes:
These pathogens may present a hazard to animal health when animals consume contaminated food, and to human health by consumption of contaminated animal products, or by direct exposure to the contaminated animal food capable of causing disease in humans.[1]
According to the FDA, “[p]athogenic E. coli are bacteria associated with foodborne illness in humans and animals.” Pathogenic E. coli bacteria “can infect animals through consumption of E. coli-contaminated animal food, and humans can be infected by handling such animal food or infected animals and their feces.”[2] The FDA has specifically addressed “[p]et foods contaminated with E. coli O157:H7,” stating that they are “of particular public health importance because they can affect both human and animal health. Pets can get sick from E. coli O157:H7 . . .and may also be carriers of the bacteria and pass it on to their human companions without appearing to be ill.”[3]
Further, the FDA’s Center for Veterinary Medicine has previously stated that the “FDA does not believe raw meat foods for animals are consistent with the goal of protecting the public from significant health risks, particularly when such products are brought into the home and/or used to feed domestic pets. . . .”[4] The FDA has advised the public that the “Federal Food, Drug, and Cosmetic Act requires that all animal foods, like human foods, be safe to eat, produced under sanitary conditions, contain no harmful substances, and be truthfully labeled.”[5]
In 2015, the FDA adopted a rule titled “Current Good Manufacturing Practice, Hazard Analysis, and Risk-Based Preventative Controls for Food for Animals.” 21 C.F.R. Part 507 (2015). For facilities that manufacture, process, pack, or hold animal food, the rule mandates, among others, that “[r]aw materials” . . . must be . . . constructed in a way that protects against contamination and deterioration, and held under conditions, e.g., appropriate temperature and relative humidity, that will minimize the potential for growth of undesirable microorganisms and prevent the animal food from becoming adulterated.” 21 C.F.R. § 507.25(b)(3).
In its recent Guidance for the Industry concerning “Hazard Analysis and Risk-Based Preventive Controls for Food for Animals,” the FDA listed pathogenic E. coli as a known or reasonably foreseeable hazard:[6]

In its Guidance, the FDA also noted that “[d]ogs and cats with foodborne illness caused by pathogenic E. coli can be asymptomatic or have symptoms ranging from mild gastroenteritis to hemorrhagic diarrhea.”[7] The FDA also referenced a “study conducted to evaluate the prevalence of microbial organisms in various types of pet food [which] found strains of non-O157:H7 Shiga toxin-producing E. coli in some raw pet food and jerky type treats (Ref. 12).”[8]
The FDA and state public health agencies “perform risk-based inspections to ensure that animal food, including pet food, is manufactured, processed, packed, and held in a manner that prevents contamination or adulteration of the pet food.”[9] When a pet food facility is not in compliance, FDA “encourages voluntary corrective actions”; when those cannot be achieved, the FDA utilizes compliance enforcement tools, including advisory actions (such as warning letters) and administrative or judicial actions.[10]
Darwin (Arrow Reliance)’s History of Repeated and Continued Violations of Food Laws
Arrow Reliance manufactures raw dog and cat food products at a facility in Tukwila, Washington. It advertises that its products use “Human-Grade Ingredients” that are “fresh, whole USDA-quality ingredients” and that such ingredients, and subsequent finished product, are “[m]inimal[ly] process[ed]” and raw.[11] Arrow Reliance emphasizes the uncooked nature of its finished product: “When we say raw, we really mean it. We do as little as possible to process your pet’s meals….”[12] Despite obvious risks of contamination, Arrow Reliance, at all relevant times, well knew that its product was frequently used in homes where small children were present, and could foreseeably be harmed from contaminated dog food.
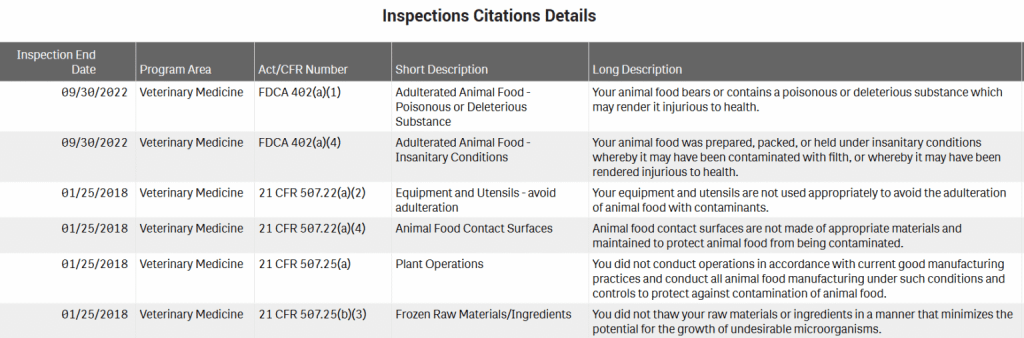
Arrow Reliance has a long history of violating pertinent food laws and regulations. To date, the FDA issued six inspection citations to the Arrow Reliance: two for violating the Federal Food, Drug and Cosmetic Act and four for violating FDA regulations. To illustrate, in 2022, the FDA found that the Arrow Reliance violated the federal legislation because its “animal food bears or contains a poisonous or deleterious substance which may render it injurious to health” and the animal food was “prepared, packed, or held under unsanitary conditions.”[13] The full list of Arrow Reliance’s citations and their descriptions follows:
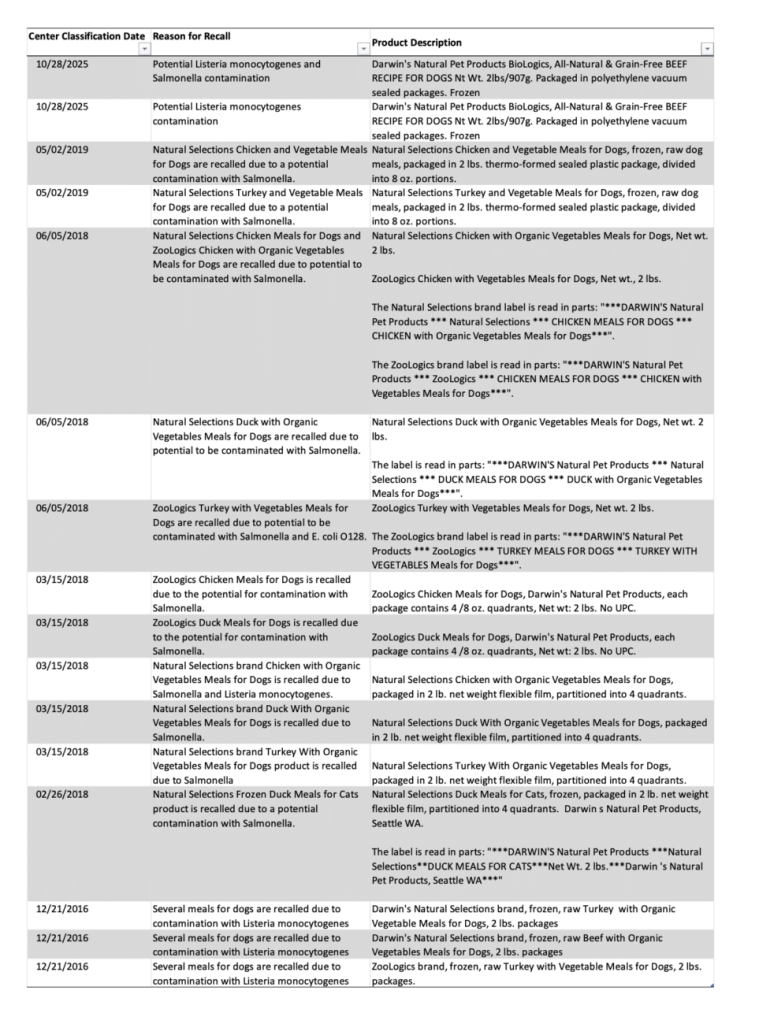
Further, Arrow Reliance’s products have been repeatedly recalled due to pathogenic contamination (Class I recalls).[14] To date, sixteen products were recalled:
The FDA issued two Warning Letters to the Arrow Reliance, one in 2018 and one in 2023. The 2018 Warning Letter noted that the FDA, during its inspections of Arrow Reliance’s facility, found that Arrow Reliance’s “raw pet food products revealed they were contaminated with Salmonella, Listeria, and/or Shiga toxin-producing Escherichia coli O128.”[15] The FDA concluded that the Arrow Reliance violated the Federal Food, Drug, and Cosmetic Act:
Your raw pet food products are food under section 201(f) of the Federal Food, Drug, and Cosmetic Act (the Act) [21 U.S.C. § 321(f)], because they are articles used for food for animals. Based on the analytical results, these products are adulterated under section 402(a)(1) of the Act, which states that food is deemed to be adulterated if it bears or contains a poisonous or deleterious substance which may render it injurious to health. The introduction or delivery for introduction into interstate commerce of an adulterated food violates section 301(a) of the Act [21 U.S.C. § 331(a)].[16]
In the 2018 Warning Letter issued to the Arrow Reliance, the FDA stated that “[t]he presence of pathogens in pet food results in pet food being adulterated when the pathogen may result in adverse health effects to animals consuming the pet food and/or humans handling the pet food.”[17] In that same letter, the FDA clearly explained that raw foods present a significant risk to human health and outlined the actions that the Arrow Reliance should take:
Unlike other human and pet foods which are heat-treated or are intended to be cooked, raw pet food has the potential to pose a significant risk to human and animal health because raw pet food is produced with minimal processing and is intended to be handled by humans and fed to animals without cooking, which would kill potentially harmful pathogens. It is therefore essential that your firm has a food safety system in place to prevent and control contamination of your products and facility. Aspects of such a system may include: proper implementation and review of cleaning and sanitization procedures, review and/or testing of your incoming materials and outgoing products, review and/or testing of your facility and environment for possible contamination, a kill step to destroy microorganism contamination, and root cause investigation and corrective action procedures when problems arise. It is your responsibility to ensure that your procedures and overall system adequately prevent, control, and respond to contamination so that you are producing an unadulterated product.[18]
The 2023 Warning Letter again found Arrow Reliance’s sampled food products, this time for cats, “to be adulterated in that they bear or contain a poisonous or deleterious substance which may render them injurious to health.”[19] In response to Arrow Reliance’s objections, the FDA reiterated in its 2023 Warning Letter that “the FDA is responsible for protecting the public (human and animal) health by ensuring, among other things, the safety of the nation’s food supply.”[20] It further stressed that the association between Salmonella-contaminated pet food and salmonellosis is well-established and that the “FDA considers a pet food to be adulterated if it is contaminated with Salmonella, regardless of the quantity or Salmonella serotype.”[21]
The FDA has also issued numerous advisories concerning Arrow Reliance’s products. As a result, it was always foreseeable to Arrow Reliance that its contaminated food posed a danger to pet owners’ families. For instance, in October 2023, FDA issued an advisory warning consumers to cease use of certain of Arrow Reliance’s raw animal food product after certain of Arrow Reliance’s product tested positive for Salmonella. In September 2024, certain of Arrow Reliance’s raw dog and cat food tested positive for Salmonella and Listeria monocytogenes. FDA advised consumers to discard those lots that tested positive for the bacteria. Arrow Reliance failed to initiate a recall for the affected product, despite FDA’s recommendation to do so, and despite the fact that transmission of pathogens from the affected product was foreseeable.
On July 29, 2025, the FDA issued an advisory concerning the product at issue here action, wherein it referenced the case of the Noah.[22] Specifically, in the July 2025 advisory, the FDA “caution[ed] pet owners that a sample of Darwin’s Natural Pet Products beef dog food made by Arrow Reliance, Inc. tested positive for Escherichia coli (E. coli) O157:H7 that is associated with a human illness.”[23] The advisory specifically referenced Noah:
In August 2024, a four-year-old child became infected with E. coli O157:H7 and subsequently developed hemolytic uremic syndrome (HUS), requiring hospitalization and extensive follow-up care. Whole genome sequencing (WGS) provided by the third-party laboratory shows that the E. coli O157:H7 present in the closed product sample of the Darwin’s beef dog food from the household freezer is a genetic match to the E. coli O157:H7 detected in a stool sample from the ill child.[24]
While the FDA recommended that Arrow Reliance recall the product lots that tested positive for E. coli O157:H7 (and Salmonella), the firm “has not recalled the affected products” to date.[25] The FDA therefore advised consumers to throw the affected products away, to not feed them to the pets, and to not donate them.[26]
Further, in the July 2025 advisory concerning the subject Arrow Reliance product, the FDA addressed why it was concerned about pathogenic contamination of pet food.
Why is the FDA concerned about E. coli O157:H7 and Salmonella in pet food?
Pet foods contaminated with E. coli O157:H7 and Salmonella are of particular public health importance because they can affect both human and animal health. Pets can get sick from E. coli O157:H7 and Salmonella and may also be carriers of the bacteria and pass it on to their human companions without appearing to be ill. In addition to the recent child case of HUS associated with pet food contaminated with E. coli O157:H7, FDA is aware of cases in which humans and/or animals have gotten sick from exposure to Salmonella-contaminated pet foods.
The Federal Food, Drug, and Cosmetic Act requires that all animal foods, like human foods, be safe to eat, produced under sanitary conditions, contain no harmful substances, and be truthfully labeled. Refrigeration or freezing does not kill E. coli O157:H7 or Salmonella.
[1] U.S. Food & Drug Admin., Biological contaminants, https://www.fda.gov/animal-veterinary/biological-chemical-and-physical-contaminants-animal-food/biological-contaminants (last visited on Mar. 30, 2026).
[2] Id.
[3] Id.
[4] U.S Food & Drug Admin., CVM Issues Assignment to Collect Official Samples of Raw Foods for Dogs or Cats in Interstate Commerce in the United States and Analyze them for Salmonella, Listeria monocytogenes, Escherichia coli O157:H7 and Non O157:H7 Shiga Toxin-Producing Escherichia coli (STEC)(June 3, 2015), https://www.fda.gov/animal-veterinary/biological-chemical-and-physical-contaminants-animal-food/cvm-issues-assignment-collect-official-samples-raw-foods-dogs-or-cats-interstate-commerce-united.
[5] U.S. Food & Drug Admin., FDA Advisory: Do Not Feed Certain Lots of Darwin’s Natural Pet Products Pet Food Due to E. coli O157:H7 and Salmonella (July 29, 2025), available at https://www.fda.gov/animal-veterinary/outbreaks-and-advisories/fda-advisory-do-not-feed-certain-lots-darwins-natural-pet-products-pet-food-due-e-coli-o157h7-and; see also U.S. Food & Drug Admin., Pet Food, https://www.fda.gov/animal-veterinary/animal-foods-feeds/pet-food (last visited on Mar. 30, 2026).
[6] U.S. Food & Drug Admin., Guidance for the Industry: Hazard Analysis and Risk-Based Preventive Controls for Food for Animals, #245 Section 3.2 (July 2022), available at https://www.fda.gov/media/110477/download?attachment.
[7] Id. at 31.
[8] Id. at 32.
[9] U.S. Food & Drug Admin., Pet Food, https://www.fda.gov/animal-veterinary/animal-foods-feeds/pet-food (last visited on Mar. 30, 2026).
[10] Id.
[12] Id.
[13] U.S. Food & Drug Admin., FDA Dashboard: Arrow Reliance, Inc., https://datadashboard.fda.gov/oii/firmprofile.htm?FEIs=3007652252&/identity/3007652252 (last visited on Mar. 30, 2026).
[14] Id.
[15] Warning Letter, Arrow Reliance, Inc., MARCS-CMS 547381, April 2, 2018 https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/arrow-reliance-inc-547381-04022018.
[16] Id.
[17] Id.
[18] Id. (emphasis added).
[19] U.S. Food & Drug Admin., Warning Letter Issued to Arrow Reliance, Inc., MARCS-CMS 644947 — February 16, 2023, https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/arrow-reliance-inc-644947-02162023.
[20] Id.
[21] Id.
[22] U.S. Food & Drug Admin., FDA Advisory: Do Not Feed Certain Lots of Darwin’s Natural Pet Products Pet Food Due to E. coli O157:H7 and Salmonella (July 29, 2025), https://www.fda.gov/animal-veterinary/outbreaks-and-advisories/fda-advisory-do-not-feed-certain-lots-darwins-natural-pet-products-pet-food-due-e-coli-o157h7-and.
[23] Id.
[24] Id.
[25] Id.
[26] Id.