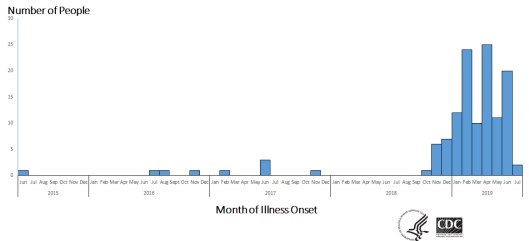
The Public Health Agency of Canada (PHAC) is investigating seven cases of Listeria monocytogenes infections in three Canadian provinces.
Five of the seven cases were reported during the period April – June 2019; the other two cases, involving the same genetic strain, occurred in 2017.
Six of the seven outbreak victims were admitted to hospital.
Outbreak cases were reported in British Columbia (1), Manitoba (1) and Ontario (5). The outbreak victims’ ages range between 51 and 97 years. Six of the seven victims are female.
 The source of the outbreak has been traced to a production batch of Rosemount brand cooked diced chicken meat, which was supplied to hotels, restaurants and institutions. The implicated product was not packaged for retail sale.
The source of the outbreak has been traced to a production batch of Rosemount brand cooked diced chicken meat, which was supplied to hotels, restaurants and institutions. The implicated product was not packaged for retail sale.
As a result of the traceback investigation, Rosemount Sales and Marketing has recalled the following product:
Rosemount brand Cooked diced chicken meat 13 mm – ½” (#16305) (4.54Kg; PACKDATE: 01/21/19; UPC 2 06 20263 12454 7)
The recalled product was supplied to hotels, restaurants and institutions in Alberta, British Columbia, Manitoba, Nova Scotia, Ontario, Quebec, and Saskatchewan, and may have been distributed nationwide.
 The fully cooked, diced chicken is a product of the USA, and was exported to Canada under USDA Export Certificate No. 039833. The manufacturer cannot be identified from the package labeling.
The fully cooked, diced chicken is a product of the USA, and was exported to Canada under USDA Export Certificate No. 039833. The manufacturer cannot be identified from the package labeling.
According to PHAC, anyone can become sick from Listeria bacteria, but those at highest risk of serious illness include pregnant women, their unborn children and newborns, adults 65 and over, and people with weakened immune systems.
Individuals living in group homes, assisted living facilities, and nursing homes would be members of one or more of these at-risk groups.
What you need to know
- Foods that are contaminated with Listeria may look, smell and taste normal. Unlike most bacteria, Listeria can survive and sometimes grow on foods being stored in the refrigerator.
- Symptoms of mild Listeria monocytogenes infections can appear as early as three days after exposure to a contaminated food, and may include fever, nausea, cramps, diarrhea, vomiting, headache, constipation, and muscle aches.
- Symptoms of severe infections may not appear until 2-3 weeks following exposure, and can take up to 70 days to develop. These symptoms can include stiff neck, confusion, headache, and loss of balance.
- Pregnant women infected with Listeria monocytogenes may experience complications including stillbirth, early delivery, and infection in the newborn baby.
- Severe cases of Listeria monocytogenes infections can be deadly.
What you need to do
- If you have Rosemount brand cooked diced chicken meat 13mm – ½” (#16305), packdate – 01/21/2019 in your food establishment, do not eat the product or serve it to others
- Secure the product and any foods made with the product in a plastic bag, throw it out and wash your hands with warm soapy water.
- If you are unsure whether your Rosemount brand chicken is part of the food recall warning, discard the product. Do not serve or consume it.
- If you suspect you have become ill from eating Rosemount brand cooked diced chicken meat, or have symptoms consistent with listeriosis, talk with your healthcare provider.