Frozen vegetables grown and processed in Egypt may be linked to a mysterious outbreak of Listeria monocytogenes infections that erupted in three stages in 2024 and 2025.
According to information obtained by eFoodAlert from the FDA in response to a Freedom of Information Act request, the outbreak was first investigated by the CDC in April 2024 without success. The investigation was reopened in August 2024 when additional cases appeared on the CDC’s radar screen. Again, the investigation was closed without a possible source having been determined.
In June 2025, the CDC reopened the investigation once more after an additional five cases attributed to the same outbreak strain were found. One of those cases occurred in December 2024; the other four from January to May 2025.
Although the outbreak comprised 27 cases overall in 13 states, according to the FDA investigation report, the CDC map provided as part of the report only shows locations for 25 of the cases: Connecticut (1), Florida (2), Illinois (1), Massachusetts (2), Maryland (1), Michigan (1), Minnesota (1), Missouri (1), New Jersey (1), New York (8), Pennsylvania (4), Rhode Island (1), and Virginia (1).
Ages of outbreak victims ranged from less than one to 97 years, with a median age of 72 years. Twenty-five of the victims were hospitalized and one person died. Two of the outbreak patients were pregnant women.
Searching for the source
The FDA did not conduct an official traceback investigation for this outbreak, as the CDC could not supply strong epidemiological evidence for a possible source. However, the agency did conduct a “limited distribution analysis” for two of the cases.
Although many of the details as to the suspected source have been redacted from the investigation report, the FOIA officials left enough breadcrumbs for eFoodAlert to follow.
The recall
The FDA collected records and samples from possible suppliers of the suspect food and, on August 20, 2025, reported positive samples with possible connection to clinical outbreak cases. Eight days later, the CDC advised the FDA that samples collected from one of the suppliers (name redacted in the investigation report supplied to eFoodAlert) was a genetic match for the outbreak strain. Samples collected from the other [unidentified] manufacturer did not match the outbreak strain.
On September 2, 2025, Endico Potatoes Inc. of Mount Vernon, NY recalled two lots of frozen vegetables due to possible Listeria monocytogenes contamination. The recall included one lot each of “frozen peas and carrots” and “mixed vegetables.” According to the Enforcement Report details, the vegetables were imported from Egypt.
In June 2024, the FDA conducted a comprehensive inspection of Endico Potatoes (Inspection ID #1232874) relating to the Foreign Supplier Verification Program. The inspection did not uncover any significant deviations or discrepancies and the results were classified as “No Action Indicated.”
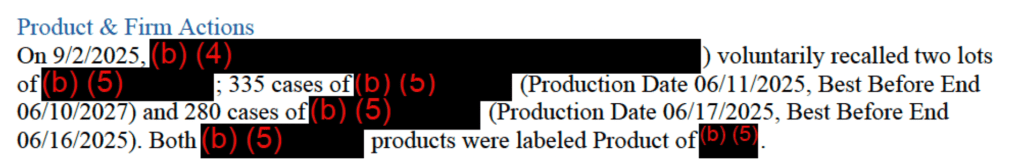
The recalled lots of frozen vegetables were manufactured after the last of the outbreak cases was detected, and the recall notice states that no illnesses were reported. Nevertheless, on September 30, 2025, the manufacturer of the frozen vegetables was added to FDA’s Import Alert 99-23, Detention Without Physical Examination of Produce Due to Contamination With Human Pathogens.
The manufacturer and supplier of the recalled vegetables was International Company For Agricultural Production & Processing (ICAPP), headquartered in Cairo, and with production facilities in Ramadan City, Ash Sharqiyah, Egypt. Both the headquarters location and the production plant site were listed in the Import Alert.
On October 22, 2025, the FDA added another supplier to Import Alert 99-23. This supplier, whose samples were positive for Listeria monocytogenes but did not match the outbreak strain, is identified in the Import Alert as Warminskie Zaklady Przetworstwa Owocowo – Warzywnego sp. o.o of Pomorskie, Poland.
Duty to inform

This would not be the first time that frozen vegetables have been linked to an outbreak of Listeria monocytogenes. In 2016, nine people were infected as a result of consuming frozen vegetables produced by CRF Frozen Foods of Pasco, Washington. A Connecticut resident died as a result of the infection, and two other outbreak victims died from other causes.
A separate outbreak between 2015 and 2018 was responsible for at least 47 illnesses and 9 deaths in Austria, Denmark, Finland, Sweden and the United Kingdom. That outbreak was traced to frozen corn and frozen vegetable mixes from a Hungarian freezing plant.
The author(s) of the FDA investigation report acknowledge that frozen vegetables are an “established vehicle” for Listeria monocytogenes, that the suspect products had an extended shelf life, and that contaminated product might still be in consumers’ homes.
The smoking gun (finding the outbreak strain in a sample of frozen vegetables) wasn’t enough for the FDA and CDC to reach a firm conclusion in this case. Instead, they identifed frozen vegetables as the “suspect vehicle” and withheld this information from the public.
Despite knowing that consumers might still be exposed to contaminated product, and despite the likelihood that frozen vegetables were the source of more than two dozen cases of Listeria monocytogenes, including one death, the FDA and the CDC remained silent.
The FDA could have issued an advisory at the time to alert consumers to the risk. This could have been done without naming the importer or the manufacturer.
The FDA should have alerted the public to this hazard, reminding consumers that frozen vegetables are not ready-to-eat products, but must be cooked thoroughly before being consumed.
Transparent opacity
The redacted FDA investigation report is an excellent example of redaction overreach.
The FOIA officer redacted information on the number of outbreak cases and their geographic distribution, even though that information was readily available in the CDC outbreak map included in the same report.
Other information that was redacted even though the information was publicly available included: the identity of the companies added to Import Alert 99-23, the details of the 2016 Listeria monocytogenes in the United States, and the details of the 2015-2018 outbreak in several European countries.
Certainly, proprietary information (e.g., product formulations and supply chain information) should be redacted as a matter of course. But, there is absolutely no justification to redacting information that is readily available to the public from other published sources.
Here is a link to the full redacted FDA Investigation Report as supplied to eFoodAlert in response to our FOIA request.
Interested in learning more about food safety and the history of foodborne disease outbreaks and investigations?
Click on the link to listen to a short excerpt, then follow the buy links to add a digital, print or audio copy to your personal library.